 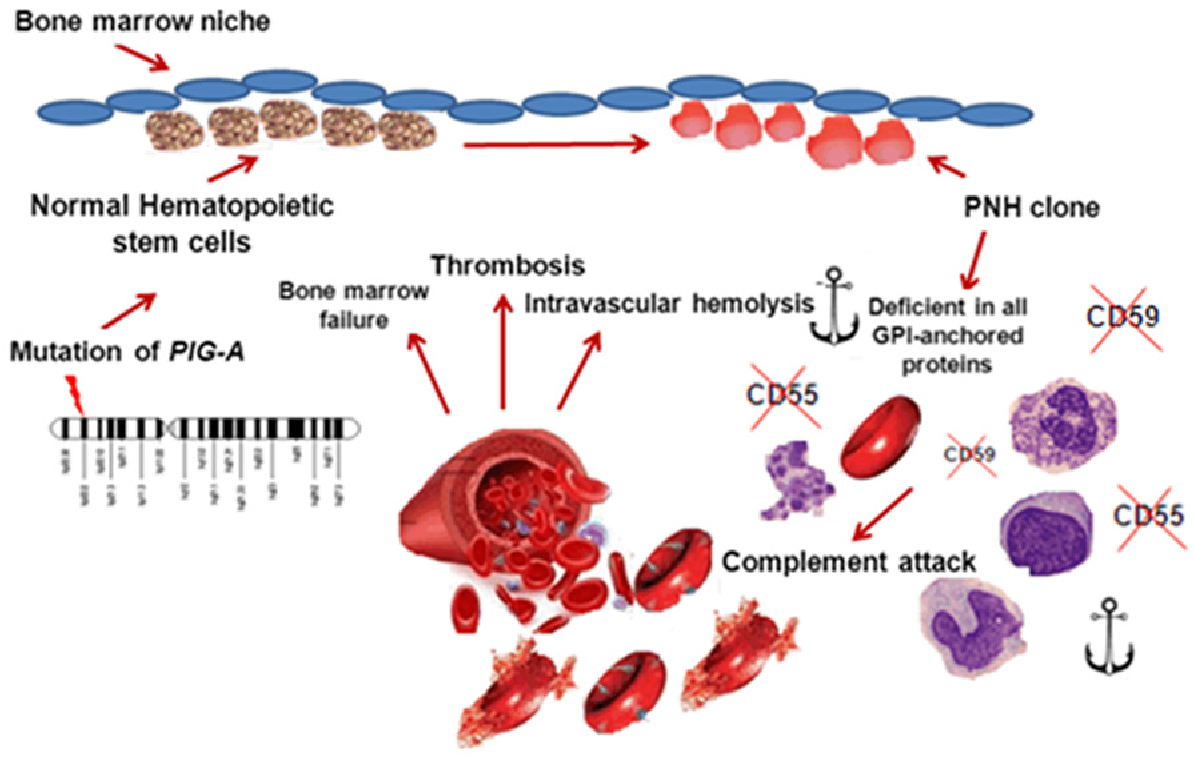
Since the complement cascade attacks the red blood cells within the blood vessels of the circulatory system, the red blood cell destruction (hemolysis) is considered an intravascular hemolytic anemia. This destructive process occurs due to deficiency of the red blood cell surface protein DAF, which normally inhibits such immune reactions. Paroxysmal nocturnal hemoglobinuria ( PNH) is a rare, acquired, life-threatening disease of the blood characterized by destruction of red blood cells by the complement system, a part of the body's innate immune system. Paroxysmal nocturnal haemoglobinuria, Marchiafava–Micheli syndrome 
In: Bone Marrow Transplantation.Medical condition Paroxysmal nocturnal hemoglobinuria doi: 10.7326/0003-4819-148-8-200804150-00003.ĭe Latour RP, Risitano AM (2020) Hemolytic processes in paroxysmal nocturnal hemoglobinuria and its treatment: intravascular and extravascular hemolysis, introduction. Narrative review: Paroxysmal nocturnal hemoglobinuria: the physiology of complement-related hemolytic anemia. Paroxysmal nocturnal haemoglobinuria (PNH) is caused by somatic mutations in the PIG-A gene. Deficiency of the GPI anchor caused by a somatic mutation of the PIG-A gene in paroxysmal nocturnal hemoglobinuria. 
Takeda J, Miyata T, Kawagoe K, Iida Y, Endo Y, Fujita T, Takahashi M, Kitani T, Kinoshita T. Overall, this study found that PNH patients receiving ECU or RAV therapy demonstrated a significant burden of illness, highlighting the need for improved PNH therapies.īurden of illness Eculizumab Fatigue Paroxysmal nocturnal hemoglobinuria Quality of life Ravulizumab.īrodsky RA. Patients reported scores below the average population norms on the Functional Assessment of Chronic Illness Therapy (FACIT)-Fatigue and European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) scales. Other patient-reported PNH symptoms included breakthrough hemolysis, shortness of breath, and headaches. Among PNH patients receiving C5i therapy for ≥ 12 months, some still reported thrombotic events (ECU, 10.0%, n = 1/10 RAV, 23.5%, n = 4/17) and required transfusions within the past year (ECU, 52.2%, n = 12/23 RAV, 22.6%, n = 7/31). A majority of patients on ECU (88.6% n = 31/35) and RAV (74.7% n = 65/87) reported fatigue symptoms. Despite most patients receiving C5i therapy for ≥ 3 months (ECU 100%, n = 35 RAV 95.4%, n = 83), many patients remained anemic with hemoglobin levels ≤ 12 g/dL in 87.5% (n = 28/32) and 82.9% (n = 68/82) of ECU and RAV recipients, respectively. This cross-sectional study surveyed 122 individuals in the USA receiving treatment for PNH with C5-targeted monoclonal antibodies, eculizumab (ECU) or ravulizumab (RAV). 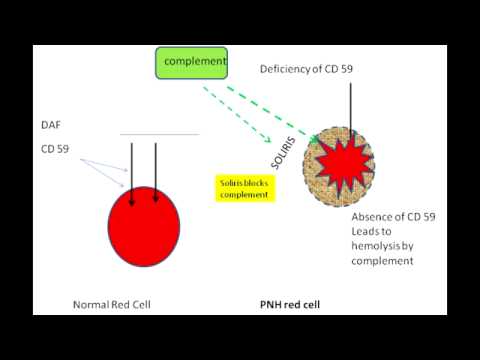
The objective of this study was to determine C5i treatment effects on clinical parameters, PNH symptoms, quality of life, and resource use for PNH patients. However, data on treatment responses and quality of life in C5-inhibitor (C5i)-treated PNH patients are scarce. Current therapies for this complement-mediated disease rely predominantly on inhibition of the C5 complement protein. Paroxysmal nocturnal hemoglobinuria (PNH) is a rare and life-threatening disease with symptoms of hemolysis and thrombosis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
